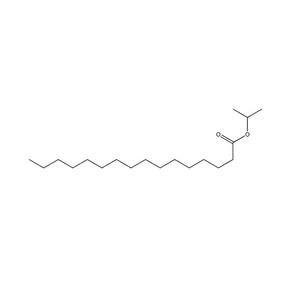
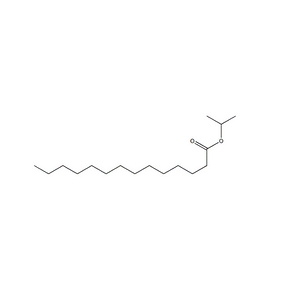
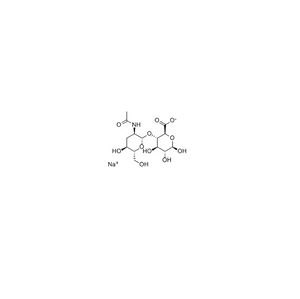
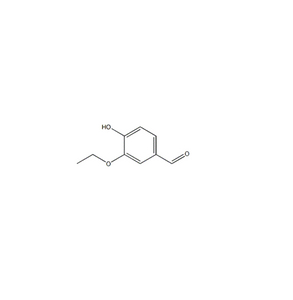
Payment & Shipping Terms | Supply Capacity |
Payment Terms: | T/T, WU | Production Capacity: | 30T/year |
Min. Order: | 1KG | Packing: | according to the customer's requirements |
Means of Transport: | Ocean, Air, Land | Delivery Date: | 7 days |
Benzoic Acid
CAS 65-85-0
Benzoic acid, also known as benzoic acid, has a molecular formula of c6h5cooh. The simplest aromatic acid with the carboxyl group directly linked to the carbon atom of the benzene ring is a compound formed by the substitution of hydrogen on the benzene ring by the carboxyl group (- COOH). It is colorless and tasteless lamellar crystal. Melting point 122.13 ℃, boiling point 249 ℃, relative density 1.2659 (15 / 4 ℃). When it sublimates rapidly at 100 ℃, its steam has a strong irritant, which is easy to cause cough after inhalation. Slightly soluble in water, easily soluble in ethanol, ether, chloroform, benzene, toluene, carbon disulfide, carbon tetrachloride, turpentine and other organic solvents. In the form of free acid, ester or its derivatives, it exists widely in nature, for example, in the form of free acid and benzyl ester in benzoin gum; it exists in some plant leaves and stems and leaves in a free form; exists in methyl oil or benzyl ester in essential oils; in horse urine, it exists in the form of its derivative, uric acid. Benzoic acid is a weak acid, stronger than fatty acid. Their chemical properties are similar, and they can form salts, esters, acyl halides, amides, anhydrides, etc., which are not easy to be oxidized. The electrophilic substitution reaction can take place on the benzene ring of benzoic acid, which is mainly produced by the intermediate substitution. Benzoic acid is often used as medicine or preservative, which can inhibit the growth of fungi, bacteria and molds. It is usually applied on the skin for the treatment of ringworm skin diseases. Used in synthetic fiber, resin, coating, rubber, tobacco industry. At first, benzoic acid was prepared by carbonization of benzoin gum or hydrolysis of alkaline water, or hydrolysis of hippuric acid. In industry, benzoic acid is produced by oxidation of toluene with air in the presence of cobalt, manganese and other catalysts, or by hydrolysis and decarboxylation of phthalic anhydride. Benzoic acid and its sodium salt can be used as bacteriostatic agent of latex, toothpaste, jam or other food, and as mordant of dyeing and printing
We can customize according to customer's requirement
If you have any question pls mail us or call us.