Payment & Shipping Terms | Supply Capacity |
Payment Terms: | T/T, WU | Production Capacity: | 500Ton/year |
Min. Order: | 1 Gram | Packing: | according to the customer's requirements |
Means of Transport: | Ocean, Air, Land | Delivery Date: | 7 days |
|
|
|
|
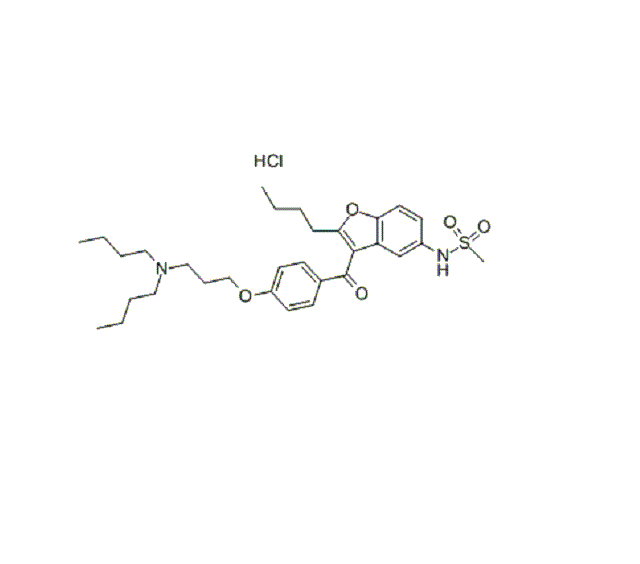
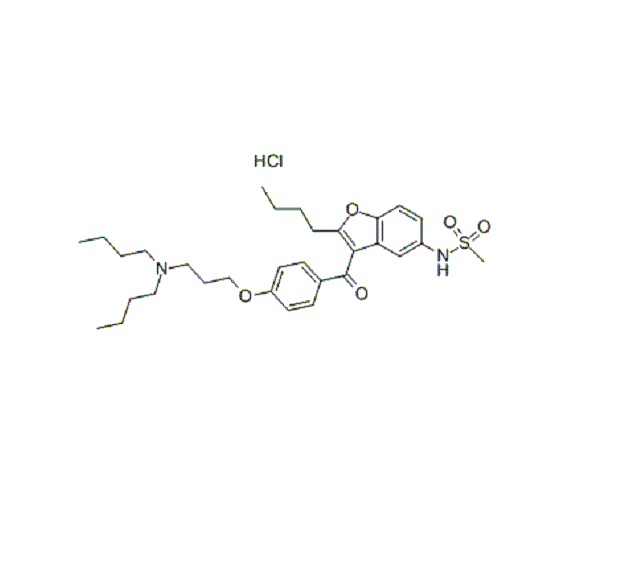
Dronedarone Hydrochloride (CAS: 141625-93-6)
Item | Index |
Molecular Formula | C31H44N2O5S.HCl |
Molecular Weight | 593.224 |
Specification | CP/USP/EP |
Appearance | White or off-white powder |
Basic Information
Dronedarone is a drug by Sanofi-Aventis, mainly for the indication of cardiac arrhythmias. It was approved by the FDA on July 2, 2009. It was recommended as an alternative to amiodarone for the treatment of atrial fibrillation and atrial flutterin people whose hearts have either returned to normal rhythm or who undergo drug therapy or electric shock treatment i.e. direct current cardioversion(DCCV) to maintain normal rhythm. In the United States, the FDA approved label includes a claim for reducing hospitalization, but not for reducing mortality, as a reduction in mortality was not demonstrated in the clinical development program. A trial of the drug in heart failure was stopped as an interim analysis showed a possible increase in heart failure deaths, in patients with moderate to severe CHF.
The U.S. label for dronedarone includes a black box warning, stating that dronedarone is contraindicated in patients with NYHA Class IV heart failure, with NYHA Class II–III heart failure with a recent decompensation requiring hospitalization or referral to a specialized heart failure clinic, or with permanent atrial fibrillation." Dronedarone is also associated with rare case of severe liver damage, including liver failure.