Payment & Shipping Terms | Supply Capacity |
Payment Terms: | T/T, WU | Production Capacity: | 600T/year |
Min. Order: | 25KG | Packing: | according to the customer's requirements |
Means of Transport: | Ocean, Air, Land | Delivery Date: | 7 days |
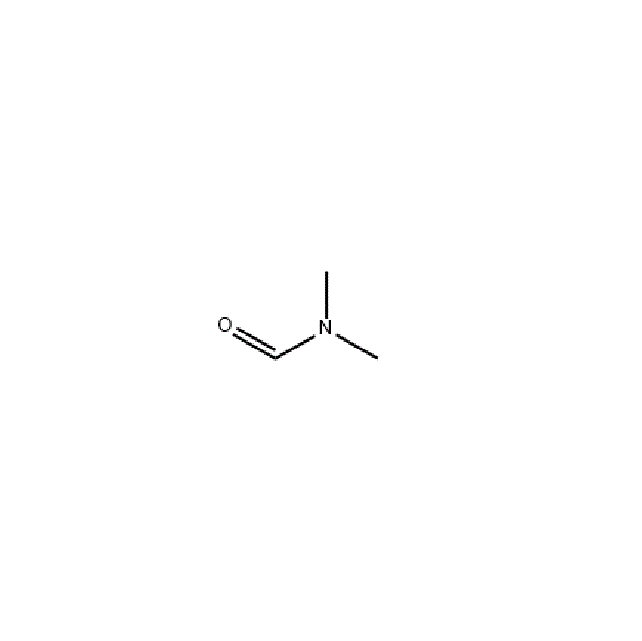
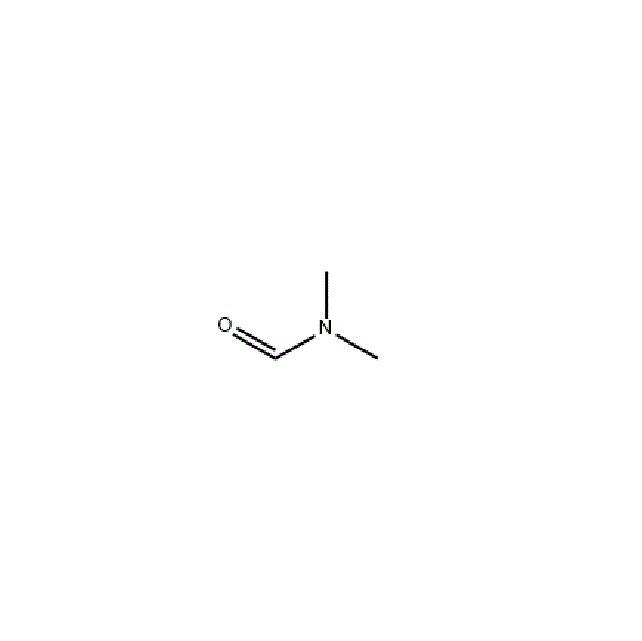
N,N-Dimethylformamide
CAS 68-12-2
Dimethylformamide is abbreviated as DMF. It is a compound produced by replacing the hydroxyl group of formic acid with a dimethylamino group, with the molecular formula HCON(CH3)2. It is a colorless, transparent, high-boiling liquid with a light amine taste and a relative density of 0.9445 (25°C). Melting point -61°C. The boiling point is 152.8°C. The flash point is 57.78°C. The vapor density is 2.51. The vapor pressure is 0.49kpa (3.7mmHg25°C). The self-ignition point is 445℃. The explosion limit of vapor and air mixture is 2.2~15.2%. Exposure to open flames and high heat can cause combustion and explosion.
Pure dimethylformamide is odorless, but industrial grade or deteriorated dimethylformamide has a fishy smell because it contains impurities of dimethylamine. The source of the name is that it is the dimethyl substituent of formamide (the amide of formic acid), and both methyl groups are located on the N (nitrogen) atom. Dimethylformamide is a polar (hydrophilic) aprotic solvent with a high boiling point, which can promote the progress of the SN2 reaction mechanism. Dimethylformamide is manufactured using formic acid and dimethylamine. Dimethylformamide is unstable in the presence of strong bases such as sodium hydroxide or strong acids such as hydrochloric acid or sulfuric acid (especially at high temperatures), and is hydrolyzed into formic acid and dimethylamine.
We can customize according to customer's requirement.
if you have any question pls mail us or call us.