Payment & Shipping Terms | Supply Capacity |
Payment Terms: | T/T, WU | Production Capacity: | 5000T/year |
Min. Order: | 25KG | Packing: | according to the customer's requirements |
Means of Transport: | Ocean, Air, Land | Delivery Date: | 7 days |
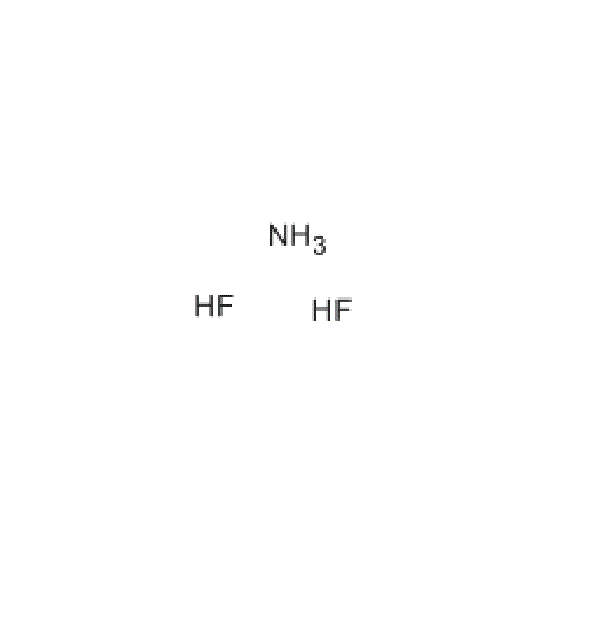
Ammonium Bifluoride
CAS: 1341-49-7
Item | Index |
Appearance | White Orthorhombic Crystal |
NH4HF2, %≥ | 98.0 |
loss on drying, %≤ | 1.0 |
Content of residue on ignition, %≤ | 0.2 |
Sulfate(base on SO4) content, %≤ | 0.1 |
Content of ammonium fluosilicate [(NH4)SiF6] , %≤ | 2.0 |
Ammonium hydrogen fluoride is the inorganic compound with the formula NH4HF2 or NH4F·HF. It is produced from ammonia and hydrogen fluoride. This colourless salt is a glass-etchant and an intermediate in a once-contemplated route to hydrofluoric acid.
Structure
Ammonium bifluoride, as its name indicates, contains a bifluoride, or hydrogen(difluoride) anion: HF2−. This centrosymmetric triatomic anion features the strongest known hydrogen bond, with a F−H length of 114 pm. and a bond energy greater than 155 kJ mol−1.
In solid [NH4][HF2], each ammonium cation is surrounded by four fluoride centers in a tetrahedron, with hydrogen-fluorine hydrogen bonds present between the hydrogen atoms of the ammonium ion and the fluorine atoms. Solutions contain tetrahedral [NH4]+ cations and linear [HF2]− anions.